Application of clinical trial inclusion criteria to clinical practice patients to quantify the burden of CNS metastases on health-related quality of life and healthcare resource use in patients with NSCLC - Lung

What Are the Documents Required for Clinical Trial Applications to Regulatory Authorities in Europe? - Sofpromed
The Clinical Trials Directive: How Is It Affecting Europe's Noncommercial Research | PLOS Clinical Trials
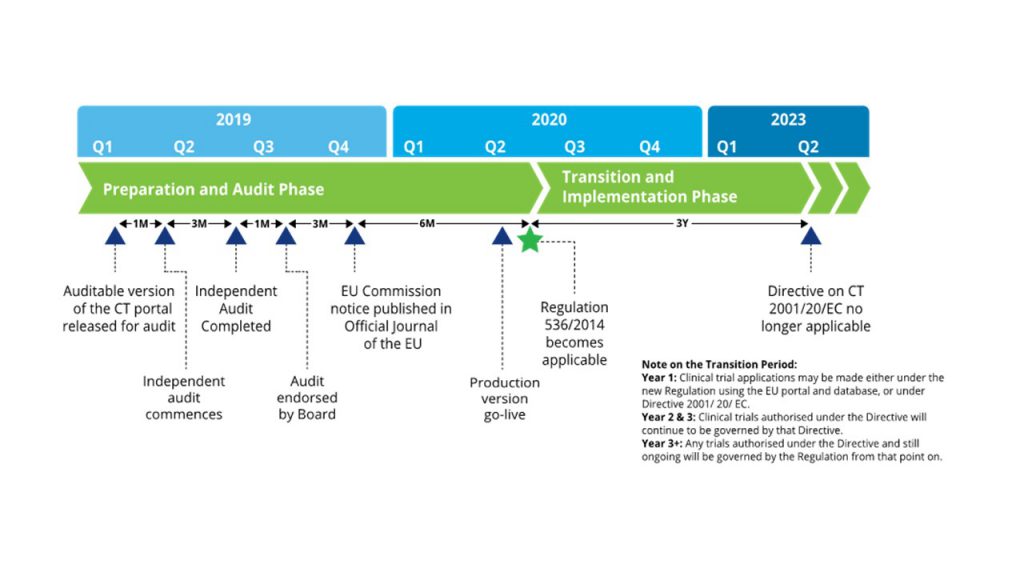
ADVANCED CLINICAL TRIALS THE CLINICAL TRIAL PROCESS: IMPENDING CHANGES IN THE REGULATORY FRAMEWORK - ADVANCED CLINICAL TRIALS
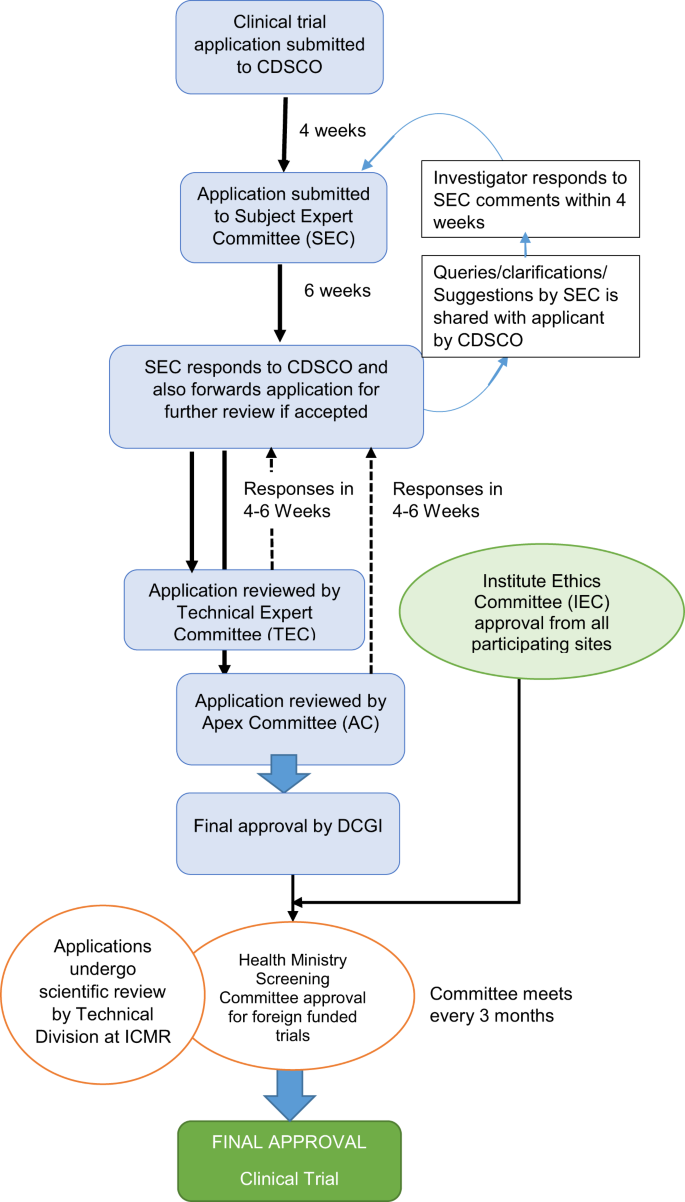
Issues, challenges, and the way forward in conducting clinical trials among neonates: investigators' perspective | Journal of Perinatology