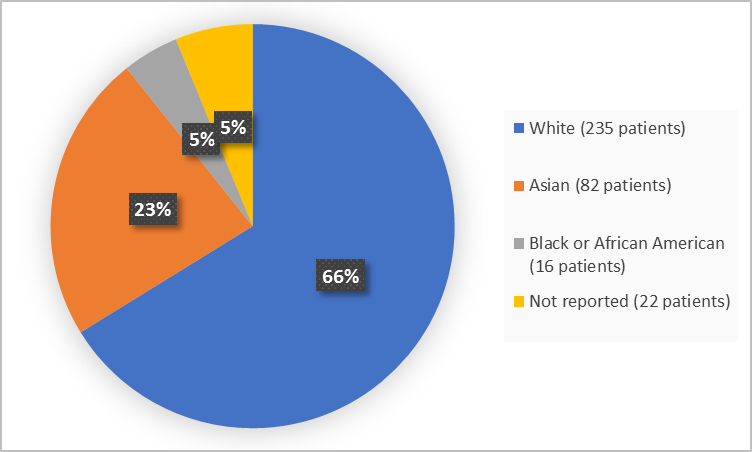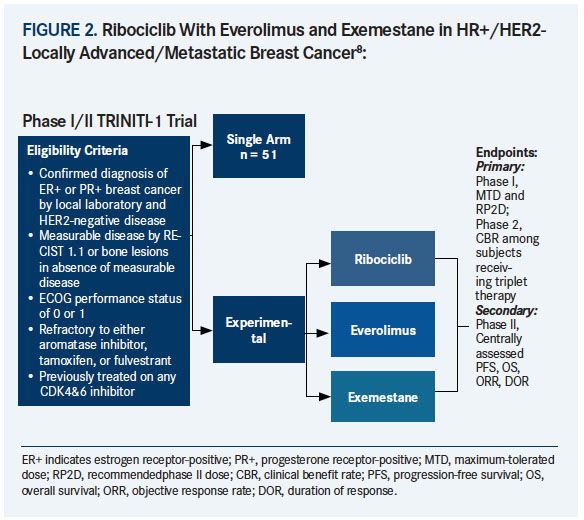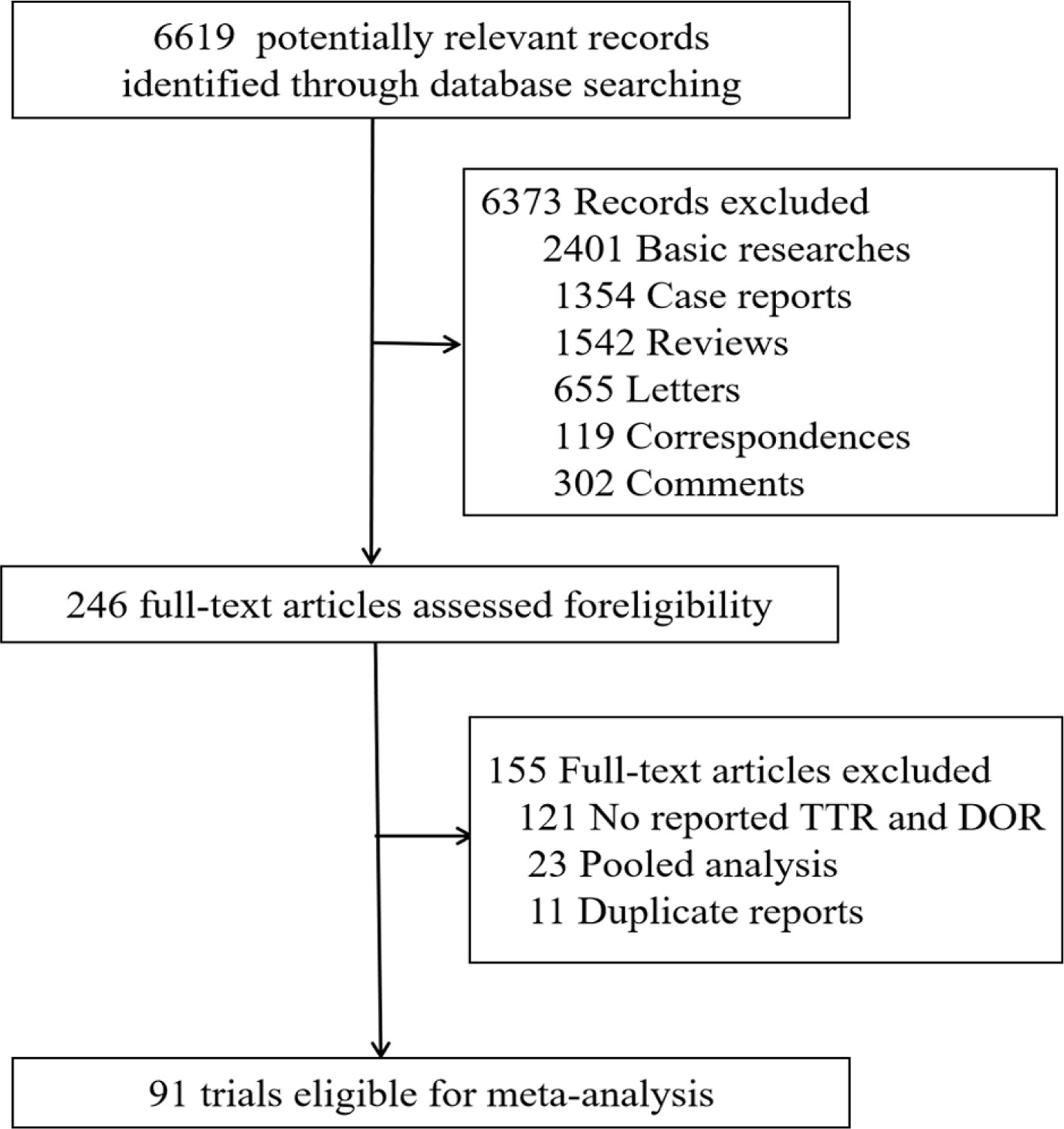
Frontiers | Response Efficacy of PD-1 and PD-L1 Inhibitors in Clinical Trials: A Systematic Review and Meta-Analysis | Oncology

Concluded and ongoing clinical trials using T-VEC in monotherapy or... | Download Scientific Diagram

Kidney Cancer - ASCO GU 2021 Highlights - Text Module - GUCS 2021 - Oncology - Clinical Care Options

Updated results from the ZUMA-5 trial of axi-cel for patients with relapsed/refractory follicular lymphoma
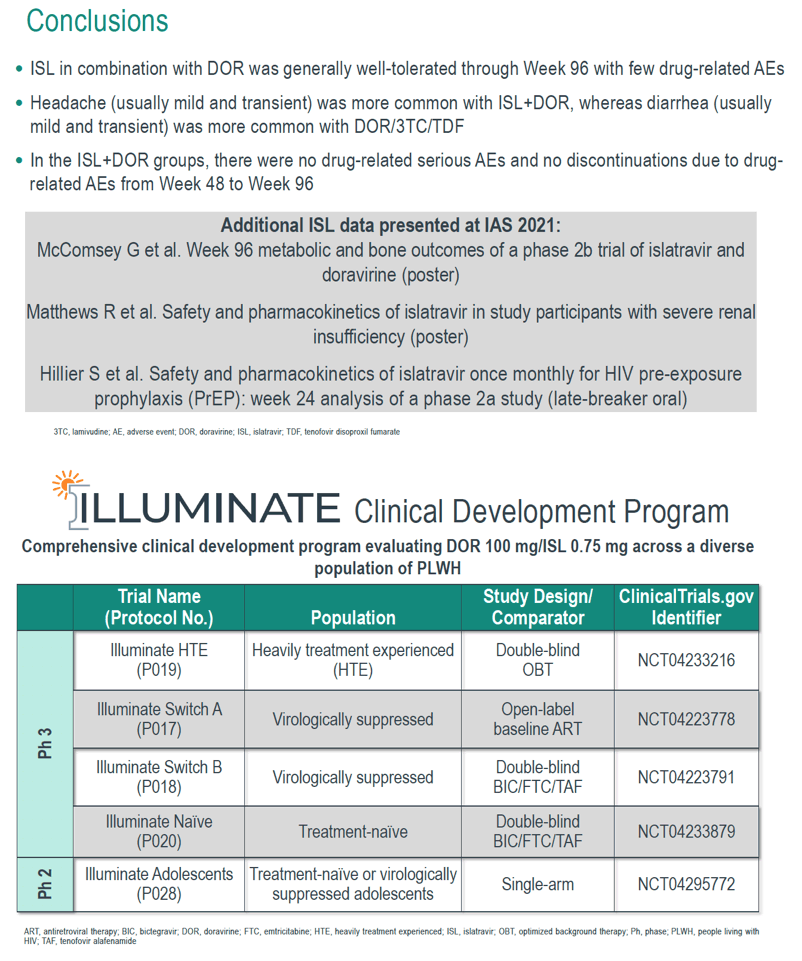
Safety Analysis of Islatravir in Combination With Doravirine in Treatment-Naïve Adults With HIV-1 Infection Through 96 Weeks Islatravir/Doravirine Adverse Events Less Frequent in Second Half of 96-Week Trial

Continued ) Summary of Genotypic and Phenotypic Resistance From DOR... | Download Scientific Diagram

A Randomized, Placebo-Controlled Trial of Pembrolizumab Plus Chemotherapy in Patients With Metastatic Squamous NSCLC: Protocol-Specified Final Analysis of KEYNOTE-407 - Journal of Thoracic Oncology