
ich e6 r1 good clinical practice consolidated guidance — Clinical Research Certification I Blog - CCRPS

The Impact of ICH GCP E6 Guideline R2 Revisions on Sponsors, Sites, Contract Research Organizations and Vendors | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services
The EMWA Budapest Working Group: A 2-year collaboration to make recommendations for aligning the ICH E3 guideline with current p

Book M1: 2022 Mini Pocket-Sized (3" x 5") ICH Guidelines for GCP (E6) – Clinical Research Resources, LLC

POL-0030: Compliance and enforcement approach and inspection strategy for clinical trials of drugs involving human subjects - Canada.ca
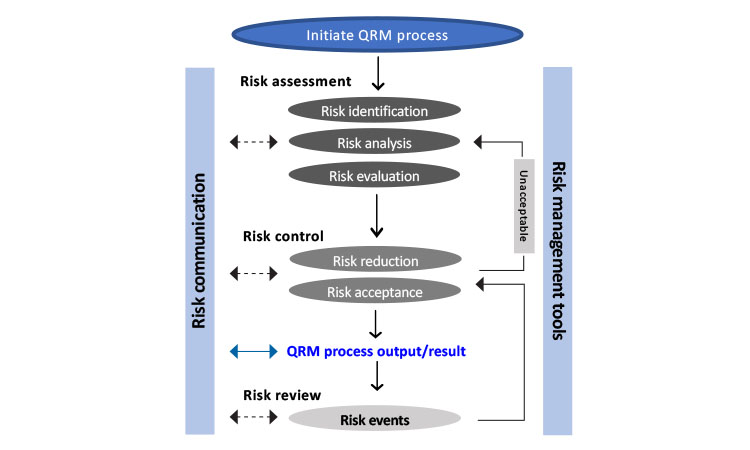
Clinical Trial Management Adaptation to ICH E6 (R2): Good Clinical Practice | Pharmaceutical Engineering










.jpg)




