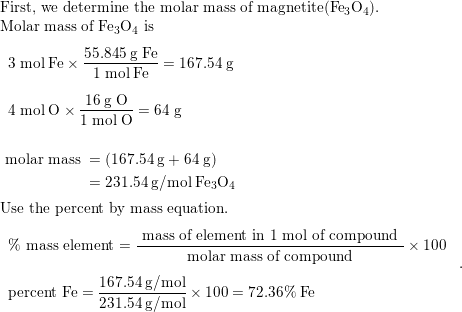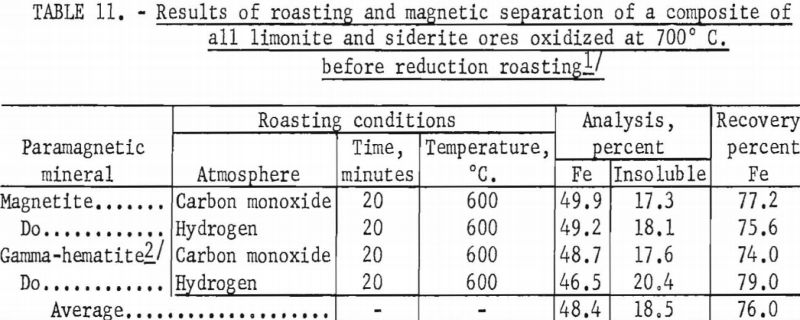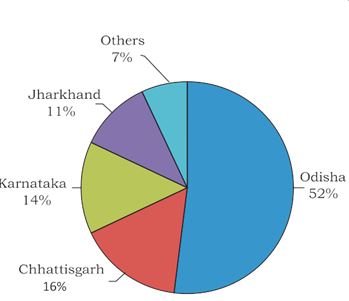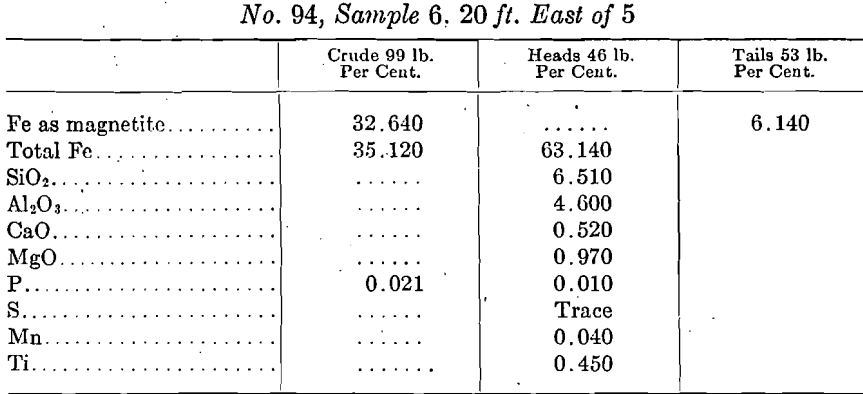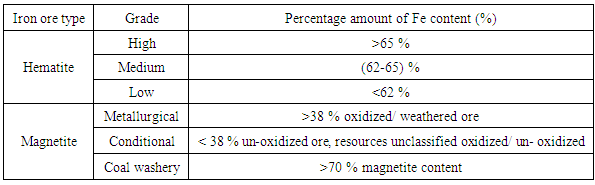
Characterization of Selected Mineral Ores in the Eastern Zone of Kenya: Case Study of Mwingi North Constituency in Kitui County

Magnetic characterisation of magnetite and hematite from the Blötberget apatite – iron oxide deposits (Bergslagen), south-central Sweden

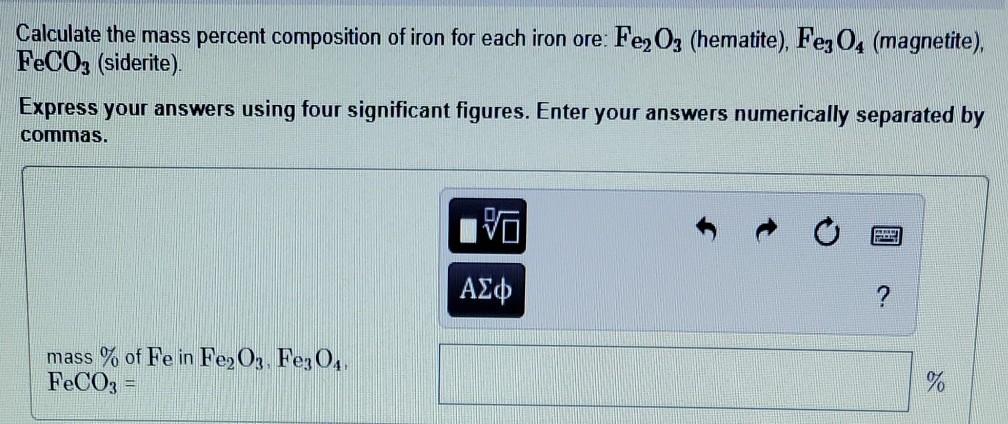
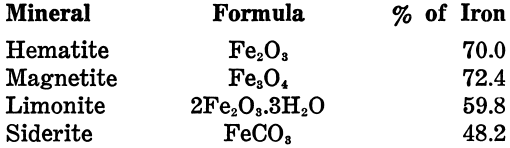
SOLVED:Iron in the earth is in the form of iron ore. Common ores include Fe2 ` O3 (hematite) , Fes C O4 (magnetite) , and FeCO3 (siderite). Calculate the mass percent composition

SOLVED:Magnetite is 4 binary compound containing only iron (55.5 gmol) and oxygen (16 g/mol) . The weight percent of iron is 72.36% . What is the empirical formula magnetite? FeO FeOz C.

Physical and magnetic properties of iron oxide nanoparticles with a different molar ratio of ferrous and ferric - ScienceDirect
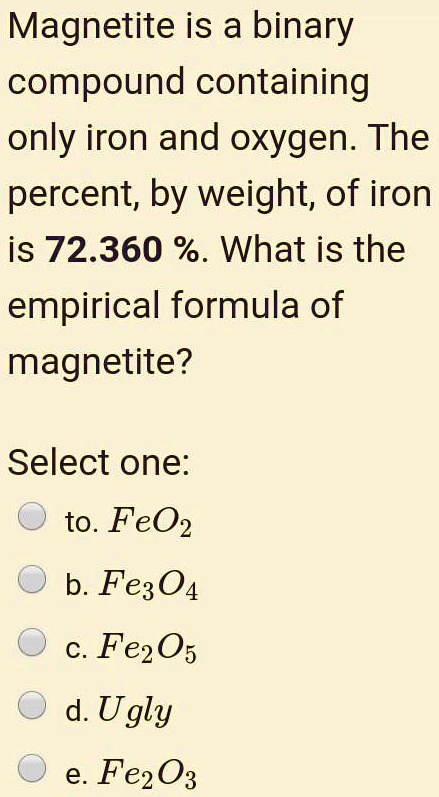
SOLVED:Magnetite is a binary compound containing only iron and oxygen: The percent; by weight; of iron is 72.360 %. What is the empirical formula of magnetite? Select one: to. FeO2 b. Fe304